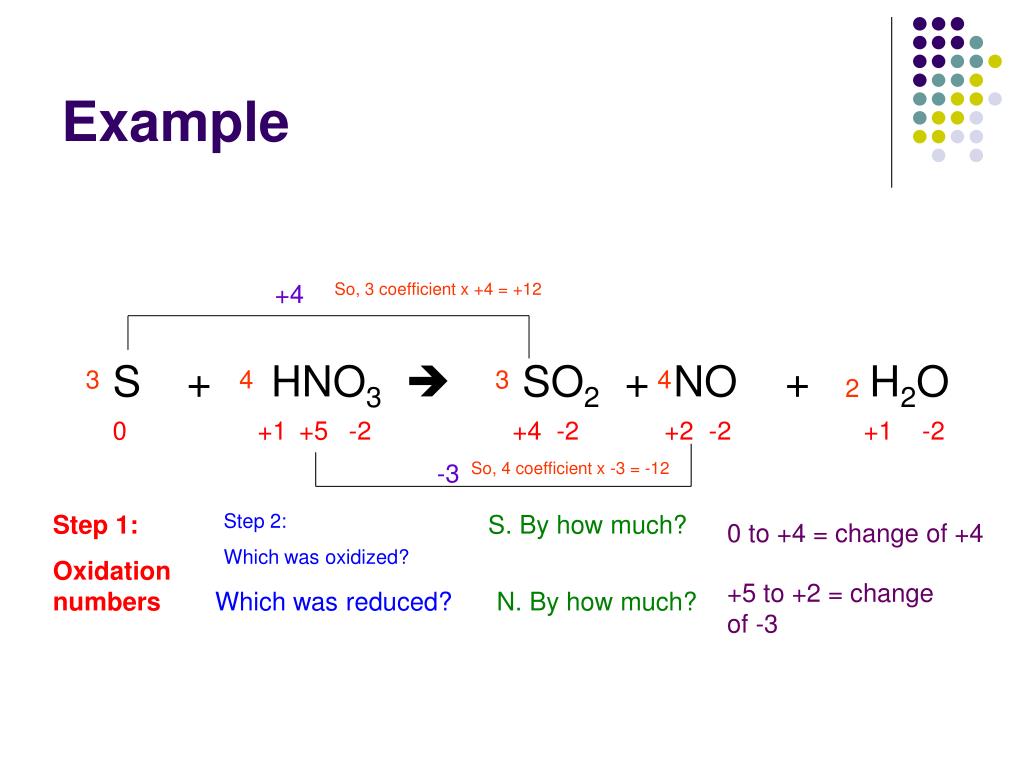
You can find online calculator of balancing redox reactions on different chemistry websites. How to Find Redox Equation Balancer?įinding a reliable redox reaction balancer is essential for accurate results. Therefore, they serve as predictive tools with some inherent limitations, and experimental validation is often necessary. Redox reaction calculators assume ideal reaction conditions and may not consider factors such as reaction kinetics, side reactions, and catalysts. While redox reactions and calculators provide valuable insights, it's important to understand their limitations. Limitations of Redox Reaction as a Predictive Tool This process involves balancing the equation to accurately represent the electron transfer and chemical changes involved. The reduction half-reaction: 4Ag+ + 4e-> 4Ag + 2H2O + 4H+ĢCu + 4AgNO3 -> 2Cu(NO3)2 + 4Ag + 2H2O + 4H+īy following these steps, you can manually calculate redox reactions. The oxidation half-reaction: 2Cu -> 2Cu2+ + 4e. Combine the balanced half-reactions to form the balanced overall redox equation.Multiply the half-reactions by appropriate integers to ensure that the number of transferred electrons is the same.Ensure that the number of electrons transferred is equal in both half-reactions.ĢAg+ + 2e-> 2Ag + H2O + 2H+ Step 6: Multiply and Combine the Half-Reactions.Balance the charges in the half-reactions by adding electrons (e-) as needed.Add hydrogen ions (H+) to balance the hydrogen atoms in both half-reactions.ĢAg+ + 2e-> 2Ag + H2O + 2H+ Step 5: Balance the Charge with Electrons.Remember to adjust the hydrogen atoms accordingly.ĢAg+ + 2e-> 2Ag + H2O Step 4: Balance Hydrogen Atoms with Hydrogen Ions.Add water molecules to balance the oxygen atoms in both half-reactions.Balance the atoms on each side of the half-reactions, except for oxygen and hydrogen atoms.ĢAg+ + 2e-> 2Ag (already balanced) Step 3: Balance Oxygen Atoms with Water Molecules.The reduction half-reaction: 2Ag+ + 2e-> 2Ag Step 2: Balance the Number of Atoms (Except Oxygen and Hydrogen) The oxidation half-reaction: Cu -> Cu2+ + 2e. Determine which species is losing electrons (oxidation) and which is gaining electrons (reduction).Separate the given equation into two half-reactions: oxidation and reduction. 
Follow below steps using an example equation Step 1: Identify the Oxidation and Reduction Half-Reactions But, one should also be familiar with manually calculating redox reactions. You can calculate redox reactions using our redox calculator. Calculating Redox Reactions manually Step-by-Step You can also use oxidation state calculator for finding oxidation number of an element or a compound. This mathematical approach helps predict and understand chemical reactions, facilitating research in diverse scientific fields. The formula relies on the conservation of mass and charge, ensuring a balanced equation representing the redox reaction. It considers the change in oxidation states of the reactants and products involved. The redox equation balancer employs a formula based on the principles of oxidation and reduction. This redox reaction calculator automates the complex calculations and provides accurate results, saving time and effort while ensuring precision. One such tool is the redox calculator, which assists chemists and students in determining the stoichiometry and balancing equations involving redox reactions. The technology has simplified the process of calculating redox reactions. Introduction to Balancing Redox Reaction Calculator It also helps them develop innovative applications in fields like environmental science, medicine, and industry. Understanding redox reactions enables scientists and chemists to comprehend the underlying mechanisms. These reactions are responsible for energy generation in batteries, corrosion of metals, synthesis of various compounds, and even the respiration process in living organisms. Redox reactions are crucial in many chemical processes due to their significance in balancing and predicting chemical equations. The Importance of Redox Reactions in Chemical Reactions These reactions involve the transfer of electrons between different chemical species, leading to changes in oxidation states.īy understanding redox reactions, we can gain insights into the transformations and interactions that occur in various chemical processes. Redox reactions, short for reduction-oxidation reactions, play a vital role in the field of chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
